

this conference is “MedTech Europe Code of Ethical Business Practice†compliant conference.
MedTech Europe Code of Ethical Business Practice

Pre-conference Workshops organized by our Scientific Partner-Turacoz are learning sessions and opportunity to discuss Biosimilars and Biologics as the newest era of pharmaceutical development. This pre-conference workshop emphasize on the understanding the trends which the biosimilar markets is heading on since its launch till the present date and moving on to the projections in future. It takes into its fold the critical developmental procedures, analyses, regulatory factors and also pharmacovigilance including post marketing surveillance and clinical studies.
Scientific Partner
Theme: Innovative and strategic approach for uptake of Biosimilars in the market
Biosimilars 2017
Theme: :
A comprehensive approach for the discussion has been taken up as listed below under several tracks and sub-tracks below:
Track 1: Current Challenges in Developing Biosimilars
The development of biologics calls for overcoming many challenges. With initial steps of concepts of biologics, their considerations, essentials for early clinical developments it is very much needed that proper scientific and strategic approaches are taken for the successful development of follow-on-biologics. Moreover, the need for overcoming the challenges continues in the late clinical steps, drug safety factors and labelling requirements. Also it is much required now to develop a drug product in accordance to Quality by Design (QbD).
This biosimilars conference will look at the facets of current challenges in biosimilar development. This biosimilar conference will focus on multiple aspects of biosimilar product development to successfully deliver safe, potential and efficacious biologic products to the market.
Related Conference:
Biosimilars Global Congress 2017, September 25-27, 2017, London, UK; World Biosimilar Congress USA 2017, May 23-24, 2017 San Diego, USA; 8th Annual Biosimilars Asia 2017, May 15-18, 2017 Shanghai, China; Biosimilars & Biobetters Congress, April 2017, London, UK, 12th Biosimilars Summit, January 24-25, 2017, Alexandria, USA; 9th Annual European Pharma Congress, July 10-12, 2017 Madrid, Spain; European Biopharma Congress, November 16-17, 2017 Vienna, Austria; 8th Asian Biosimilars Congress, August 10-12, 2017 Beijing, China
BioSim-Asociación Española de Biosimilares , Medicines for Europe(EGA) , GPhA-Generic Pharmaceutical Association , IGBA- International Generic and Biosimilars Medicines Association , BGMA-British Biosimilars Association
Track 2: Emerging Biosimilars in Therapeutics
The explorations in the field of biologics have created a new avenue for the clinicians towards better disease management. The emerging biologics have already manifested fruitful outcomes in treatment of ailments like those of psoriasis, rheumatic arthritis, certain cancers, inflammatory bowel disease(IBD) etc .Emerging Biosimilar insulins are likely to enter the insulin landscape as patents for major branded insulin products start to expire in the next few years. The main theme of this track is to have sound knowledge in the emerging biosimilars like Filgrastim, Pegfilgrastim, Recombinant blood products, therapeutic proteins, vaccines, Biosimilar anti-bodies, Growth hormones, Biosimilar peptides, therapeutic proteins and other biosimilar developments and their impacts.
Related Conference:
Biosimilars Global Congress 2017, September 25-27, 2017, London, UK; World Biosimilar Congress USA 2017, May 23-24, 2017 San Diego, USA; 8th Annual Biosimilars Asia 2017, May 15-18, 2017 Shanghai, China; Biosimilars & Biobetters Congress, April 2017, London, UK, 9th Annual European Pharma Congress, July 10-12, 2017 Madrid, Spain; European Biopharma Congress, November 16-17, 2017 Vienna, Austria; 8th Asian Biosimilars Congress, August 10-12, 2017 Beijing, China
IGBA- International Generic and Biosimilars Medicines Association , BGMA-British Biosimilars Association, BioSim-Asociación Española de Biosimilares, Medicines for Europe(EGA), GPhA-Generic Pharmaceutical Association
Track 3: Biosimilars Analytical Strategies
Analysis of biosimilars and biologics forms to be one of the most important aspect towards the biologics and biosimilar development process. Biosimilars analytical methods for process development and validation as well as use of production technologies such as disposables and supply chain logistics can help companies establish facility flexibility.
This biosimilars global event also includes Bioanalytical methods, Formulation, Bioassay for comparability and potency testing, GMP protein analysis, LC/MS analysis for discovery, preclinical, and clinical programs.
Related Conferences:
12th Biosimilars Summit, January 24-25, 2017, Alexandria, USA; World Biosimilar Congress USA 2017, May 23-24, 2017 San Diego, USA; 8th Annual Biosimilars Asia 2017, May 15-18, 2017 Shanghai, China; ;Biosimilars Global Congress 2017, September 25-27, 2017, London, UK; Biosimilars & Biobetters Congress, April 2017, London, UK; 9th Annual European Pharma Congress, July 10-12, 2017 Madrid, Spain; European Biopharma Congress, November 16-17, 2017 Vienna, Austria; 8th Asian Biosimilars Congress, August 10-12, 2017 Beijing, China
BGMA-British Biosimilars Association, BioSim-Asociación Española de Biosimilares, GPhA-Generic Pharmaceutical Association, IGBA- International Generic and Biosimilars Medicines Association, Medicines for Europe(EGA)
Track 4: Regulatory Approach for Biosimilars
Biosimilars are the generic version of biological. It is the new buzz word in pharmaceutical industry. Biosimilars are highly similar to licensed reference product not withstanding minor differences in clinically inactive components; also there are no clinically meaningful differences between the biologicals and the reference product in terms of safety, purity, and potency. This track includes: Licensing of biosimilars, Biosimilars regulation, Patent issues, BLA filing for biosimilars, Biosimilars regulatory prospects of BRIC countries, a paradigm of traditional generics to Biosimilars , Biowaiver approval for Biosimilars and other aspects of Biosimilar approvals. Biosimilars 2017 will provide an excellent and global opportunity to the scientists, partners and pharma leaders from Biopharmaceutical and Biotechnology industries to innovate and to explore the strategic market for Biosimilars and Biologics with a clear picture of the regulatory approach for biosimilars and biologics.
Related Conference:
12th Biosimilars Summit, January 24-25, 2017, Alexandria, USA; World Biosimilar Congress USA 2017, May 23-24, 2017 San Diego, USA; 8th Annual Biosimilars Asia 2017, May 15-18, 2017 Shanghai, China; ;Biosimilars Global Congress 2017, September 25-27, 2017, London, UK; Biosimilars & Biobetters Congress, April 2017, London, UK; 9th Annual European Pharma Congress, July 10-12, 2017 Madrid, Spain; European Biopharma Congress, November 16-17, 2017 Vienna, Austria; 8th Asian Biosimilars Congress, August 10-12, 2017 Beijing, China
IGBA- International Generic and Biosimilars Medicines Association, Medicines for Europe(EGA), BGMA-British Biosimilars Association, BioSim-Asociación Española de Biosimilares
Track 5: Legal Issues and BPCI Act
The Patient Protection and Affordable Care Act (PPAC Act) was signed into law in March 2010 in addition to the amendments in the Public Health Service Act (PHS Act) to create an abbreviated approval pathway for biosimilars and follow on biologics. These new statutory provisions are often referred as the Biologics Price Competition and Innovation Act of 2009 (BPCI Act). This track concentrates upon such legal bindings and the aspects of the BPCI Act that pertain to the biosimilars and biologics. This session on legal issues shall be very beneficial to research scientists from both academic backgrounds and also those from industry R&D. The Europe biosimilars, US biosimilars, Australian biosimilars, Canada origin biosimilars and UK biosimilars are expected to take control over the biosimilar market around the globe by 2017.
Related Conference:
Biosimilars Global Congress 2017, September 25-27, 2017, London, UK; World Biosimilar Congress USA 2017, May 23-24, 2017 San Diego, USA; 8th Annual Biosimilars Asia 2017, May 15-18, 2017 Shanghai, China; Biosimilars & Biobetters Congress, April 2017, London, UK, 12th Biosimilars Summit, January 24-25, 2017, Alexandria, USA; 9th Annual European Pharma Congress, July 10-12, 2017 Madrid, Spain; European Biopharma Congress, November 16-17, 2017 Vienna, Austria; 8th Asian Biosimilars Congress, August 10-12, 2017 Beijing, China
BioSim-Asociación Española de Biosimilares , Medicines for Europe(EGA) , GPhA-Generic Pharmaceutical Association , IGBA- International Generic and Biosimilars Medicines Association , BGMA-British Biosimilars Association
Track 6: Biosimilars Research Pipeline
Biosimilars is a biologic medical product which is copy of an original product that is manufactured by a different company. There are some specific scientific consideration for criteria, design and analysis regarding development of Biosimilars. Biosimilars are officially approved versions of original biosimilar innovator products, and can be manufactured when the original product's patent expires.
This session shall be highly beneficial for the biosimilar industry researchers to update themselves on the latest research updates from around the world. This session also finds place for all the biosimilar exhibitions associated with the field of biosimilars and biologics.
Related Conference:
Biosimilars Global Congress 2017, September 25-27, 2017, London, UK; World Biosimilar Congress USA 2017, May 23-24, 2017 San Diego, USA; 8th Annual Biosimilars Asia 2017, May 15-18, 2017 Shanghai, China; Biosimilars & Biobetters Congress, April 2017, London, UK, 12th Biosimilars Summit, January 24-25, 2017, Alexandria, USA; 9th Annual European Pharma Congress, July 10-12, 2017 Madrid, Spain; European Biopharma Congress, November 16-17, 2017 Vienna, Austria; 8th Asian Biosimilars Congress, August 10-12, 2017 Beijing, China
GPhA-Generic Pharmaceutical Association, BioSim-Asociación Española de Biosimilares, Medicines for Europe(EGA
Track 7: Globalization of Biosimilars
This track discuses about the generic drugs impact on global biosimilar market and Cost and risk management, Adopting innovative mechanisms such as risk-sharing arrangement, LATAM market for biosimilars.
Those who can attend the biosimilar meeting under this track are ones following Biologics/ Proteins/Biosimilar Products, New Biosimilar Development, Process Science, Biosimilar Market, Portfolio Management, Research & Development, Business Development, Business Operations and Scientific Affairs.
Related Conference:
Biosimilars Global Congress 2017, September 25-27, 2017, London, UK; World Biosimilar Congress USA 2017, May 23-24, 2017 San Diego, USA; 8th Annual Biosimilars Asia 2017, May 15-18, 2017 Shanghai, China; Biosimilars & Biobetters Congress, April 2017, London, UK, 12th Biosimilars Summit, January 24-25, 2017, Alexandria, USA; 9th Annual European Pharma Congress, July 10-12, 2017 Madrid, Spain; European Biopharma Congress, November 16-17, 2017 Vienna, Austria; 8th Asian Biosimilars Congress, August 10-12, 2017 Beijing, China
BGMA-British Biosimilars Association, BioSim-Asociación Española de Biosimilares, GPhA-Generic Pharmaceutical Association, IGBA- International Generic and Biosimilars Medicines Association, Medicines for Europe(EGA)
Track 8: Clinical Development Of Biosimilars
This track includes Clinical trials on major diseases Risk management, and quality affairs, Case studies, and clinical models, Transgenic animals, Targeted cell line development, PK/PD studies, Toxicological studies, ethics maintained in clinical and preclinical studies, development difficulties and Aspects of genotoxicity tests. This track is designed for those who are having sound knowledge on clinical studies and clinicians prospects for biosimilars. Biosimilar guidelines on the above mentioned topics are also to be thrown light upon at this biosimilars conference.
Related Conference:
Biosimilars Global Congress 2017, September 25-27, 2017, London, UK; World Biosimilar Congress USA 2017, May 23-24, 2017 San Diego, USA; 8th Annual Biosimilars Asia 2017, May 15-18, 2017 Shanghai, China; Biosimilars & Biobetters Congress, April 2017, London, UK, 12th Biosimilars Summit, January 24-25, 2017, Alexandria, USA; 9th Annual European Pharma Congress, July 10-12, 2017 Madrid, Spain; European Biopharma Congress, November 16-17, 2017 Vienna, Austria; 8th Asian Biosimilars Congress, August 10-12, 2017 Beijing, China
BioSim-Asociación Española de Biosimilares, IGBA- International Generic and Biosimilars Medicines Association, Medicines for Europe(EGA), BGMA-British Biosimilars Association
Track 9: Intellectual Property Rights
Needless to mention differentially that any pharma product has to successfully pass through the regulatory affairs' scanners before being launched in the market , Biosimilars and biologics are no exceptions. Apart from regulatory aspects the companies developing these biosimilars and biologics tend to enjoy monopoly over their product. Intellectual Property Rights (IPR) and patents are ultimate tools to the biosimilar manufacturers for safeguarding their interests. 2016 was a record year for development of Biosimilars & biologics in US. In this year 16 IPR petition was filed. So, expected scenario of 2017 would be a boom in Biosimilar and biologics field.
Pharma patent scenario, Criteria for patentability and patent claims, Patentable subject matter, biosimilar guidelines, biosimilar patent issues are key points of discussion in the segment. Patent analysts, law firms and legal authorities dealing with biosimilar regulatory affairs would find this conference a perfect platform to put forward their views on the topic.
Related Conference:
Biosimilars Global Congress 2017, September 25-27, 2017, London, UK; World Biosimilar Congress USA 2017, May 23-24, 2017 San Diego, USA; 8th Annual Biosimilars Asia 2017, May 15-18, 2017 Shanghai, China; Biosimilars & Biobetters Congress, April 2017, London, UK, 12th Biosimilars Summit, January 24-25, 2017, Alexandria, USA; 9th Annual European Pharma Congress, July 10-12, 2017 Madrid, Spain; European Biopharma Congress, November 16-17, 2017 Vienna, Austria; 8th Asian Biosimilars Congress, August 10-12, 2017 Beijing, China
IGBA- International Generic and Biosimilars Medicines Association , BGMA-British Biosimilars Association, BioSim-Asociación Española de Biosimilares, Medicines for Europe(EGA), GPhA-Generic Pharmaceutical Association
Track 10: Bioequivalence Assessment
Bioequivalence focuses on the equivalence of release of the active pharmaceutical ingredient from the pharmaceutical product and its subsequent absorption into the systemic circulation. This also includes Strategies for the bioequivalence assessment of topical dosage forms, Bioequivalence assessment of respiratory dosage forms, Bioequivalence approaches for transdermal dosage forms, stastical criteria for establishing biosimilarity and interchangeability of follow on-biologics. It also highlights the scientific issues & considerations regarding the assessment of Biosimilars and biologics, exposure and response of bioequivalence assessment to support the release of Biosimilars to the market.
Related Conference:
Biosimilars Global Congress 2017, September 25-27, 2017, London, UK; World Biosimilar Congress USA 2017, May 23-24, 2017 San Diego, USA; 8th Annual Biosimilars Asia 2017, May 15-18, 2017 Shanghai, China; Biosimilars & Biobetters Congress, April 2017, London, UK, 12th Biosimilars Summit, January 24-25, 2017, Alexandria, USA; 9th Annual European Pharma Congress, July 10-12, 2017 Madrid, Spain; European Biopharma Congress, November 16-17, 2017 Vienna, Austria; 8th Asian Biosimilars Congress, August 10-12, 2017 Beijing, China
BioSim-Asociación Española de Biosimilares , Medicines for Europe(EGA) , GPhA-Generic Pharmaceutical Association , IGBA- International Generic and Biosimilars Medicines Association , BGMA-British Biosimilars Association
Track 11: BCS and IVIVC Based Biowaivers
Biopharmaceutics Classification System and invitro and invivo classification discusses about ADME pathways of different drugs. This also includes BCS biowaivers, In vitro diffusion cells for dissolution testing in formulation development, In vitro preclinical ADME/BCS testing. The objective of this work was to suggest the biowaivers potential of biopharmaceutical classification system which are known to increase the solubility, dissolution, oral absorption of water insoluble drugs. The Biosimilars Congress 2017 aims at addressing all such challenges of the pharma formulation sector for biobetters, biologics, biosimilars and biowaivers.
Excellences in the field of drug development, drug design, biotechnology, Biopharmaceutics will find their place in this track.
Related Conference:
Biosimilars Global Congress 2017, September 25-27, 2017, London, UK; World Biosimilar Congress USA 2017, May 23-24, 2017 San Diego, USA; 8th Annual Biosimilars Asia 2017, May 15-18, 2017 Shanghai, China; Biosimilars & Biobetters Congress, April 2017, London, UK, 12th Biosimilars Summit, January 24-25, 2017, Alexandria, USA; 9th Annual European Pharma Congress, July 10-12, 2017 Madrid, Spain; European Biopharma Congress, November 16-17, 2017 Vienna, Austria; 8th Asian Biosimilars Congress, August 10-12, 2017 Beijing, China
IGBA- International Generic and Biosimilars Medicines Association , BGMA-British Biosimilars Association, BioSim-Asociación Española de Biosimilares, Medicines for Europe(EGA), GPhA-Generic Pharmaceutical Association
Track 12: Biosimilar Companies and Market Analysis
This track is concentrated towards the different reviews and forecasts regarding the scenario of Biosimilars market and follow on Biologics. The present status and future scenario of the market are best to be discussed during this session. Market researches from the first launching of biosimilar to the newest one till date prospects for a radical change in the pharmaceutical market. Market researchers, market analysts, industrialists would be the apt participants for this session at Biosimilars 2017.
Related Conference:
12th Biosimilars Summit, January 24-25, 2017, Alexandria, USA; World Biosimilar Congress USA 2017, May 23-24, 2017 San Diego, USA; 8th Annual Biosimilars Asia 2017, May 15-18, 2017 Shanghai, China; ;Biosimilars Global Congress 2017, September 25-27, 2017, London, UK; Biosimilars & Biobetters Congress, April 2017, London, UK; 9th Annual European Pharma Congress, July 10-12, 2017 Madrid, Spain; European Biopharma Congress, November 16-17, 2017 Vienna, Austria; 8th Asian Biosimilars Congress, August 10-12, 2017 Beijing, China
Track 13: Challenges in Biosimilars Pharmacovigilance
This session of the Biosimilars 2017 looks into the future and FDA initiatives that have already been announced to include enhanced tracking and follow-up of post market issues, planned improvements in AERS, and pilots of new post market drug-monitoring strategies. Current challenges in pharmacovigilance, Adverse drug reactions with pharmaceutical products, Biosimilar guidelines for pharmacovigilance practice and pharmacoepidemiology are the points that shall be laid emphasis in this session.
Related Conference:
12th Biosimilars Summit, January 24-25, 2017, Alexandria, USA; World Biosimilar Congress USA 2017, May 23-24, 2017 San Diego, USA; 8th Annual Biosimilars Asia 2017, May 15-18, 2017 Shanghai, China; ;Biosimilars Global Congress 2017, September 25-27, 2017, London, UK; Biosimilars & Biobetters Congress, April 2017, London, UK; 9th Annual European Pharma Congress, July 10-12, 2017 Madrid, Spain; European Biopharma Congress, November 16-17, 2017 Vienna, Austria; 8th Asian Biosimilars Congress, August 10-12, 2017 Beijing, China
IGBA- International Generic and Biosimilars Medicines Association, Medicines for Europe(EGA), BGMA-British Biosimilars Association, BioSim-Asociación Española de Biosimilares
Track 14: Brexit Effect on Biosimilars
The Brexit effect on Biosimilars tends to be negative. Not only it would be a major setback towards approval and launch of biosimilars to the market but also be hindrance towards the cost cutting approach taken up by NHS.With Britain being among principal clinical trial centers is owned to see a decrease in the willingness of the manufacturers and researchers to carry out any further trials in Britain. Also Brexit will cause the principal motive of Bristish Biosimilars Association(BBA) to fall back- which aimed at increasing the use of Biosimilars.
Track 15: Entrepreneurs Investment Meet
Entrepreneurs who are willing to put in hard work and invest in the field of biologics and biosimilars will find this meeting the best place to properly shape their drive for the new endeavours. Also this meeting will help them find the best experts who can make their investment fruitful and worthwhile. The best technological knowhow, economical aspects, regulatory red tapes and profit shares involving biosimilars and biologics shall be discussed at this Biosimilars Global Event.
The Biosimilars conference 2017 is owned to bring together the worldwide top pharmaceuticals, biotechnology and regulatory representatives under one roof that will provide them the best platform to update and share knowledge related to biosimilars and biologics. Hence, the International Biosimilar Conference will unfold the facets of biosimilars, ranging from the evolving regulatory landscapes, biosimilar guidelines to the legal and economic aspects and the then current challenges in biosimilar development. This biosimilar conference will focus on a variety of aspects of biosimilar product development to successful delivery of safe, efficacious and potent biosimilar products to the market. By attending this biologics conference one will gain a comprehensive outlook on the key issues surrounding biosimilars. An important platform for Biosimilars stakeholders to discuss and share best practices in expediting development in the field of Biologics and Biosimilars is ought to be Biosimilars 2017
Why to attend???
Join your peers around the world focused on learning about Biologics and Biosimilars related advances, which is your single best opportunity to reach the largest assemblage of participants from the Biosimilars community, conduct demonstrations, distribute information, meet with current and potential professionals, make a splash with a new research works, and receive name recognition at this 3-day event. World-renowned speakers, the most recent research, advances, and the newest updates in Biologics and Biosimilars are hallmarks of this conference.
Target Audience:
- Students, Scientists, Researchers, and Faculty of Pharmaceutical Sciences
- Universities, Medical Colleges, Researchers from Pharmaceutical backgrounds
- Pharma Industry, Pharmacy Associations and Societies,
- Business Entrepreneurs, Training Institutes, Software developing companies,
- Medical Devices Manufacturing Companies, CRO
- Data Management Companies.
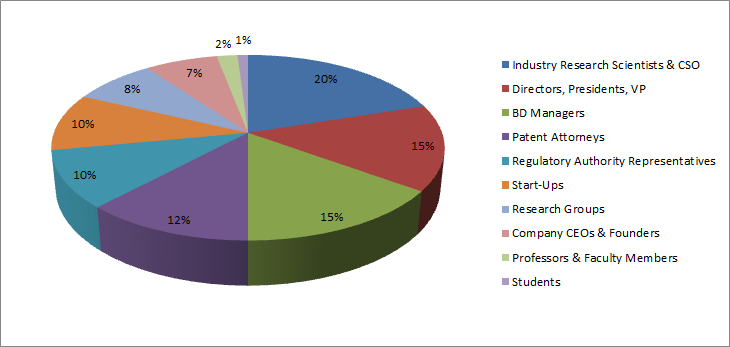
Past Conference Attendee Share
For precise details about the conference:
As per market researchers, the global biosimilar market is expected to be worth $24 billion in 2019-at compound annual growth rate (CAGR) of some 65% from 2014.Clinical trials at global level would incur an approximate cost of few hundred crores.The estimated cost of developing biosimilars for global markets is $75-250 million, while developing traditional non-biologic generics costs about $2-3 million. With the introduction of Inflectra on April 06, 2016 the Biosimilar Market will grow at even higher rate.
Critical Analysis:
The capital-intensive nature of the biosimilar business and long gestation periods between initial investment
and commercialization require biosimilar players to take a higher level of risk than their counterparts in the smallmolecule generics business, and this can be a significant barrier to entry for many start-ups.
At 2021 the global market of biosimilars is envisage to reach USD 10.90 Billion. Previously it was USD 3.39 Billion in 2016. From 2015 to 2020 the global biosimlar market expected to reach CAGR of 49.1%. This opinion covers knowledge on biosimilars that are prepared for market purpose in obedience with the European Medical Association, U.S. FDA and other regulatory authorities. Extreme factors behind this growth are escalating requirement for bio similar drugs because of their worthwhileness, increasing old aged people, pressure to lessen the spending’s on health care, and high expenditure on biologics.
Segments: Oncology, auto immune diseases, infectious diseases, diabetes mellitus, glycoprotein hormone diseases, endocrine disorders and other some other applications are segments of biosimilars. By considering this all of this oncology is envisage to reach most CAGR during further period.
Regions: According to region, biosimilars can be divided into North America Europe, and Asia. Important members of the market of biosimilars comprise Hospira, Amgen, Teva, Sandoz International GMBH, Dr. Reddy’s Lab., Biocon, Celltrion, Catalent, Mylan and Merck.
Over the previous several years, biologics have improved momentous traction in the pharmaceutical industry, illustrating beyond $150 billion in global sales in 2013. And hope to generate $290 billion in revenue constitute 27% of the pharmaceutical market by 2020. 48% of sales arrive from 11 biologics that aspect loss of individuality. Analysts envisage the worldwide market of biosimilars by 2020 to grasp $25-$35 billion. Since the biosimilars approval in the EU in 2006, there are at present more than 700 biosimilars accepted world widely. In dominant markets like the European Union, regulators and payers have perceived the probable financial asset of biosimilars over the mentioned products. In the U.S alone, the cost preserving from diverting to biosimilars are expected to be between $40v and $250 billion over the following 10 years, with the 1st biosimilars to shot the US market project about $5-7billion in preserving.
Biological drugs consists similar clinical properties in points of assurance, capability, efficacy, and pureness by analyzed to the authentic biologic drug. Global market of biosimilars is predicted to developed at their rapid pace as blockbuster biologics are accessing patent termination; and thus , and construct enormous potential for the comparable biological medicinal products. The U.S. and APAC markets present beneficial growth opportunities for the related biological medicinal products industry, which is predicted to develop at a momentous rate in the coming years.

Brand name biologic are as worth as about $100,000 per patient. Biosimilars on the contrary can be afford at a 30-40% curtailed than that of the brand name product. However considering all the achieving stories and opportunities there also lies a significant 50% failure rate in progressing and achieving towards marketing of biosimilars.
The U.S. holds the biggest share of the global biologics market value and produces half of the share value advancement as well. The European Union accounts for 22% of the sales on market value and 14% of its value growth. In contrast, arriving markets perform just a silver of the sales pie, which contains 7.5% share. Thus, biologics market advancement is still broadly consumed by sophisticated markets. The global biologics market had attains $170 billion in sales figure in 2012, occupying 18% of the overall market. The 5 top 10 global products in terms of sales volume are biologic’s; on 2008 it was two only.
Roughly twenty percent of the biologics and biosimilars in the global R&D pipelines are facing challenges to fall in line with the regulatory instructions.
This term of the Biosimilars will look into the forth coming days and FDA drive that have already been declared to having enlarged tracking and follow-up of post marketing examination problems, prepared improvements in AERS, and pilots of new post market drug-monitoring action and ADR relavant problems. Biosimilar instructions for pharmacovigilance practice and pharmacoepidemiology are the marks that shall be grab attention in this session.
The global Biosimilars market is envisage to reach $26.5Billion by 2020 increasing at a CAGR of 49.1% from 2015 to 2020 whereas The global biosimilars market alone is nearly to reach $6.22 Billion by 2020 from $2.29 Billion in 2015, at a compound annual growth rate (CAGR) of 22.1% from 2015 to 2020.
The global biosimilars market is influenced by Europe, pursued by Asia-Pacific, Rest of the World (Row), and North America. However, the Asia-Pacific region is likely to witness the highest growth rate during the projection period.
The global biosimilar market is increasing at an epidemic rate. The CAGR from 2015 to 2020 is predicted at over 22%. The biosimilar market is habitual to be around $6.2 billion by 2020 from only $2.3 billion in 2015. By the end of this decade the biosimilar would confidently cover 27% of the total pharmaceutical market. Moreover, with the global rise in concern for more accessible-developed- cost friendly healthcare, biosimilar drugs would be a relevant choice to the payers, end users, manufacturers over the valuable reference biologics.
Conference Highlights:
Major Pharma Associations around the Globe
- Academy of Managed Care Pharmacy (AMCP), Alexandria, VA
- American Association of Pakistani Pharmaceutical Scientists
- British Columbia Pharmacy Association
- California Pharmacists Association
- Kansas Pharmacists Association
- Ohio Pharmacists Association
- Pharmacy Guild of Australia
- World Health Organization (WHO)
Target Audience:
- Professors, Associate Professors, Assistant Professors
- PhD Scholars
- Graduates and Post Graduates
- Directors, CEO’s of Organizations
- Association, Association presidents and professionals
- Noble laureates in Health Care and Medicine
- Bio instruments Professionals
- Bio-informatics Professionals
- Software development companies
- Research Institutes and members
- Supply Chain companies
- Manufacturing Companies
- CRO and DATA management Companies
- Training Institutes
- Business Entrepreneurs
Audience Share:
- Industry 60%
- Academia 30%
- Others 10%
Pre-conference Workshops are organized by our Scientific Partner-Turacoz and are quality learning sessions and opportunity to discuss in brief on Biosimilars and Biologics - the newest buzz in pharmaceutical development. Thess pre-conference workshops emphasize on the understanding the trends which the biosimilar markets is heading on since its launch till the present date and moving on to the projections in future. It takes into its fold the critical developmental procedures, analyses, regulatory factors and also pharmacovigilance including post marketing surveillance and clinical studies.
The 10th International Conference and Exhibition on Biologics and Biosimilars will provide an opportunity to hear and meet Researchers, Practitioners, Students, Clinicians, and Business Professionals to share expertise, foster collaborations and assess rising innovations across the world. This will be equally informative for participants who attend this meeting as well as a for key opinion leaders who want to brush up on their knowledge outside their own area of specialization.
In association with Turacoz Ltd. we look forward towards arranging a great Biosimilars and biologics expert networking meet and knowledge exchange sessions at USA this October.
For more details contact Kiara Dcruize at biosimilars@pharmaceuticalconferences.org.
Upcoming Pre-conference Workshop:
A few glimpses of the past Pre-Conference Workshops:




Conference Highlights
- Current Challenges in Developing Biosimilars
- Emerging Biosimilars in Therapeutics
- Biosimilars Analytical Strategies
- Regulatory Approach for Biosimilars
- Legal Issues and BPCI Act
- Biosimilars Research Pipeline
- Globalization of Biosimilars
- Clinical Development Of Biosimilars
- Intellectual Property Rights
- Bioequivalence Assessment
- BCS and IVIVC Based Biowaivers
- Biosimilar Companies and Market Analysis
- Challenges in Biosimilars Pharmacovigilance
- Brexit Effect on Biosimilars
- Entrepreneurs Investment Meet
To share your views and research, please click here to register for the Conference.
To Collaborate Scientific Professionals around the World
| Conference Date | October 16-17, 2017 | ||
| Sponsors & Exhibitors |
|
||
| Speaker Opportunity Closed | Day 1 | Day 2 | |
| Poster Opportunity Closed | Click Here to View | ||
Useful Links
Special Issues
All accepted abstracts will be published in respective Our International Journals.
- Journal of Bioanalysis & Biomedicine
- Journal of Bioequivalence & Bioavailability
- Journal of Pharmaceutical Sciences & Emerging Drugs
Abstracts will be provided with Digital Object Identifier by



























